 
This is based on comparing the structure with. Formal charges for all the different atoms. The oxygen has a formal charge of 6 - 4 - 2 = 0 (same ordering of terms). Formal charge group number of atom of interest - electrons in the circle of atom of interest. This video is useful for students taking general. 
The central carbon has a formal charge of 4 (valence electrons) - 0 (lone pairs) - 4 (bonds) = 0. This chemistry video tutorial provides a basic introduction into how to calculate the formal charge of an atom or element in a lewis structure. Formal charges should be as small as possible (comparing the absolute value. We can double-check formal charge calculations by determining the sum of the formal charges for the whole structure. The sum of the formal charges must equal to the total charge on the molecule or ion. O, N, and C are in groups 6, 5, and 4 and their. All atoms in the molecule have zero formal charge, the "happiest" situation for any molecule. We calculate the formal charge of an atom in a molecule or polyatomic ions as follows: Formal Charge (valence electrons of the 'free' element) - (unshared electrons) - (bonds). Draw a reasonable Lewis dot structure for ONC, including the formal charges (N.B. However for calculating the formal charge partial atomic charge concept is not required, it is first to see the valency (how many bonds are connected to the. As bonds or charges are added or removed. Each hydrogen atom has one electron as a neutral atom, no lone pairs and shares one bond, for a formal charge of zero. In MarvinSketch, the charge of an atom is initially set to be neutral. It has no lone pairs, and it shares four bonds, so the formal charge is zero. Formal charge equation formally compares the number of valence electrons in an isolated neutral atom (which can be determined from the older style group number. The carbon in CH 4 has four electrons as a neutral atom. Subtract the number of bonds shared by the atom.Subtract the number of non-bonding electrons (usually in lone pairs).Count the number of valence electrons of the neutral atom.The sum of the formal charges, with a couple of extra rules, will help us to decide which of multiple-possible valid Lewis structures is likely to be the correct one. Formal charge is just a way of bookkeeping that helps us to decide which of multiple Lewis structures is the likely true bonding arrangement of a covalent molecule. The formal charges can be calculated using the formula given below: The formal charge of an atom valence electrons of an atom non-bonding electrons ½ (bonding electrons) The valence electrons (V.E) of an atom are the total number of electrons present in its valence shell. Electron in the bonded atom dots around the atom + lines connected. The 100 free and reliable online calculators that help you to solve any calculation-related problems and provides you with the. In those cases we resort to calculating what's called the formal charge of each atom. Formal charge valence electron in the unbonded atom - electron in the bonded atom. Also, it places the least electronegative atom in the center, and the negative charge on the more electronegative element (Guideline 4).Sometimes it's difficult to tell which of two possible Lewis structures of a compound represents the actual bonding of the molecule. However, the first arrangement of atoms is preferred because it has the lowest number of atoms with nonzero formal charges (Guideline 2). 
Note that the sum of the formal charges in each case is equal to the charge of the ion (–1). 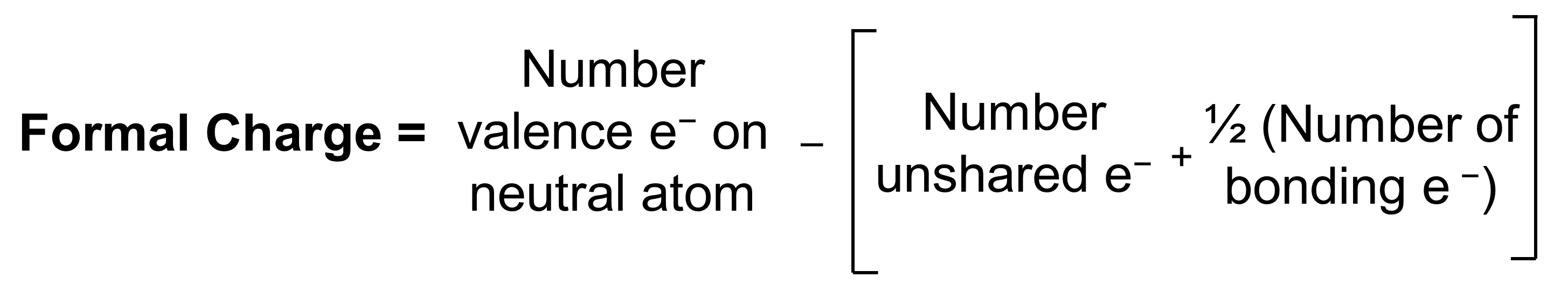
Possible Lewis structures and the formal charges for each of the three possible structures for the thiocyanate ion are shown here: This charge is assigned based on the assumption that electrons are distributed equally inside a molecule. The formal charges present in each of these molecular structures can help us pick the most likely arrangement of atoms. For example, if we want to obtain the Lewis structure of the Sulfate ion, SO4 2, we must first enter the charge by typing (-2) or by entering -2 in the charge. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
